
These periodic tables use accurate data for name, atomic number, element symbol, atomic weight, and electron. In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is. Some tables are available as slides in Google Apps. In general, atoms are most stable, least. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons. The valence electrons are the electrons in the outermost electron shell of an atom. For non-transition metals, we count to 8, but for transition metals, we count to 12. 
Looking at the orbitals explains how valence electrons work for transition metals. Up to four dots are placed above, below, to. To write an element’s Lewis dot symbol, we place dots representing its valence electrons, one at a time, around the element’s chemical symbol. consists of the chemical symbol for the element plus a dot for each valence electron. For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). allows you to keep track of valence electrons during bond formation. The shells after (ignoring transition metals) represent the s and p-orbitals. So what does this have to do with our shells? The first "shell" represents the 1s orbital. Valence electrons are outer shell electrons for main group elements. For example, carbon has an electron configuration of 1s 22s 22p 2. In chemistry and physics, a valence electron is an electron associated with an atom that can form a chemical bond and participate in a chemical reactions. The way we count our electrons is by moving from right to left, starting at the beginning of the table. 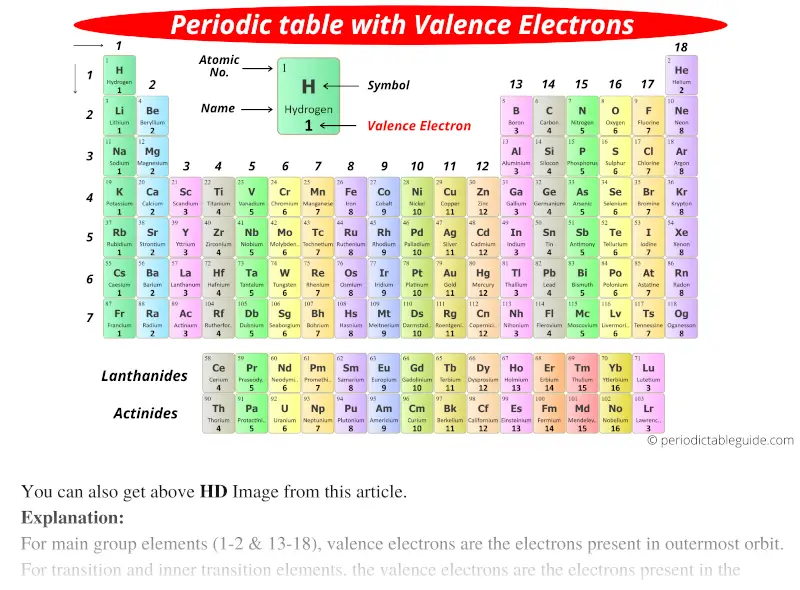
F-orbitals start appearing in the lanthanides and actinides (the separated two rows). P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). In the second period elements, the two electrons in the 1 s sublevel are called inner-shell electrons and are not involved directly in the element's reactivity, or in the formation of compounds. Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level. Valence electrons are the electrons in the highest occupied principal energy level of an atom.Valence is denoted using a positive or negative integer used to. This is determined based on the number of electrons that would be added, lost, or shared if it reacts with other atoms. Valence describes how easily an atom or radical can combine with other chemical species. Has 5 subshells, each holding 2 electrons, for a total of 10 electrons The words valence and valency have two related meanings in chemistry.Introduced in 1868, the term is used to express both the power of combination of an element in general and the numerical value of the power of combination. Has 3 subshells, each holding 2 electrons, for a total of 6 electrons Valence, in chemistry, the property of an element that determines the number of other atoms with which an atom of the element can combine.** Except for helium, which has only two valence electrons. 
* The general method for counting valence electrons is generally not useful for transition metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
